| Successful modification | The parasite was generated by the genetic modification |
| The mutant contains the following genetic modification(s) |
Gene disruption,
Introduction of a transgene
|
| Reference (PubMed-PMID number) |
Reference 1 (PMID number) : 24509910
|
| MR4 number |
|
| top of page |
| Parent parasite used to introduce the genetic modification |
| Rodent Malaria Parasite | P. berghei |
| Parent strain/line | P. berghei ANKA |
| Name parent line/clone |
P. berghei ANKA 676m1cl1 (RMgm-29)
|
| Other information parent line | 676m1cl1 (RMgm-29) is a reference ANKA mutant line which expresses GFP-luciferase under control of a constitutive promoter. This reference line does not contain a drug-selectable marker (PubMed: PMID: 16242190). |
| top of page |
| The mutant parasite was generated by |
| Name PI/Researcher | T. Annoura; S.M. Khan; C.J. Janse |
| Name Group/Department | Leiden Malaria Research Group |
| Name Institute | Leiden University Medical Center (LUMC) |
| City | Leiden |
| Country | The Netherlands |
| top of page |
| Name of the mutant parasite |
| RMgm number | RMgm-930 |
| Principal name | 1424cl1 |
| Alternative name | Δsequestrin-a |
| Standardized name | |
| Is the mutant parasite cloned after genetic modification | Yes |
| top of page |
| Phenotype |
| Asexual blood stage | Not different from wild type |
| Gametocyte/Gamete | Not different from wild type |
| Fertilization and ookinete | Not different from wild type |
| Oocyst | Not different from wild type |
| Sporozoite | Not different from wild type |
| Liver stage | Mutant sporozoites showed normal gliding motility and WT-levels of hepatocyte invasion. Mice infected with either 1 or 5x104 PbΔsequestrin sporozoites, intravenously, had a 2-3 day delay in blood-stage patency when compared to WT sporozoites infections and 4 out of 11 mice did not develop a blood-stage infection after inoculation with 1x104 sporozoites. These observations show that liver stage development is strongly affected in the absence of sequestrin and the 2-3 day prolonged prepatent period is indicative of a >99% reduction in liver-stage development. PbΔsequestrin liver stages have normal morphology, with respect to cell division, size and PVM formation at 24hpi. However at 48hpi, as determined by staining with anti-MSP1 antibodies, all liver-stage parasites were MSP1 negative. To investigate the maturation of these parasites, 54hpi parasites were examined using anti-MSP1 and anti-EXP1 antibodies. Over 60% WT parasites at this time point were strongly MSP1 positive, whereas the majority of PbΔsequestrin parasites were MSP1 negative, with only around 7% of parasites exhibiting similar MSP1 staining. |
| Additional remarks phenotype | Mutant/mutation
The mutant lacks expression of sequestrin/lisp2 (liver stage specific protein 2; LISP2) and expresses the fusion gene GFP-Luciferase under the control of the constitutive eefa promoter
Protein (function)
PBANKA_100300 encodes a 2172 amino acid protein with a large repeat region of approximately 1000 amino acids. This region is composed of several repeat motifs, particularly 12 copies of a 56-amino acid motif. Analyses of the amino acid sequence with various structure analysis tools indicates that it has an N-terminal signal sequence but no other motifs for membrane association such as transmembrane domains or a GPI-anchor motif, suggesting that the encoded product has the structure of a secreted protein. It contains a predicted Plasmodium 6-cysteine motif. Several studies analysing expression of this protein in P. berghei (see 'Additional mutants') provide evidence for specific expression in liver stages. The protein has been named LISP2 and sequestrin.
Phenotype
Mutant sporozoites showed normal gliding motility and WT-levels of hepatocyte invasion. Mice infected with either 1 or 5x104 PbΔsequestrin sporozoites, intravenously, had a 2-3 day delay in blood-stage patency when compared to WT sporozoites infections and 4 out of 11 mice did not develop a blood-stage infection after inoculation with 1x104 sporozoites.
These observations show that liver stage development is strongly affected in the absence of sequestrin and the 2-3 day prolonged prepatent period is indicative of a >99% reduction in liver-stage development.
PbΔsequestrin liver stages have normal morphology, with respect to cell division, size and PVM formation at 24hpi. However at 48hpi, as determined by staining with anti-MSP1 antibodies, all liver-stage parasites were MSP1 negative. To investigate the maturation of these parasites, 54hpi parasites were examined using anti-MSP1 and anti-EXP1 antibodies. Over 60% WT parasites at this time point were strongly MSP1 positive, whereas the majority of PbΔsequestrin parasites were MSP1 negative, with only around 7% of parasites exhibiting similar MSP1 staining.
Imaging of mice infected with 1x104 PbΔsequestrin sporozoites, showed a strong reduction in luminescence signals (>10-fold reduction) compared to signals found in mice infected with the same number of WT-luciferase expressing sporozoites.
Combined these observations show that sequestrin plays a role in late liver stage development which is in agreement with observations made by Orito et al. (RMgm-799) who show a role for sequestrin during late liver stage development and show that mutants lacking sequestrin have a 30-100 fold reduction in liver stage development.
Additional information
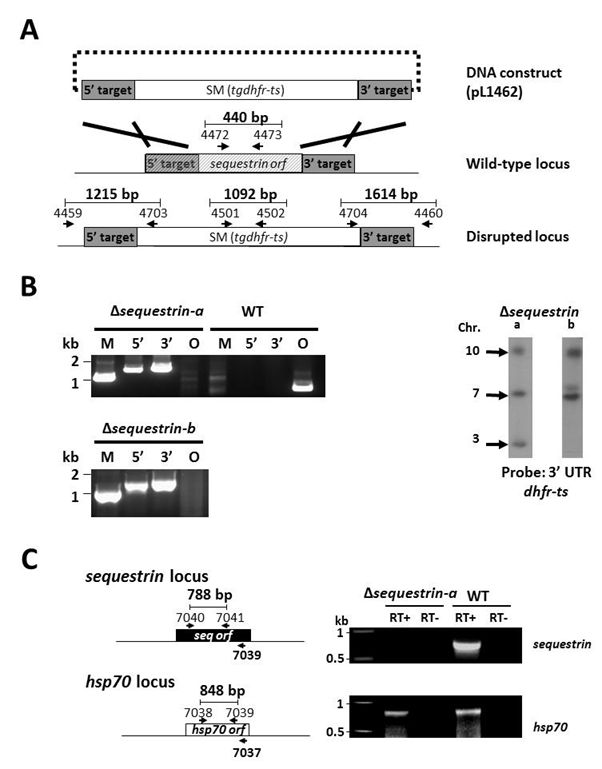
Supporting Figure S4
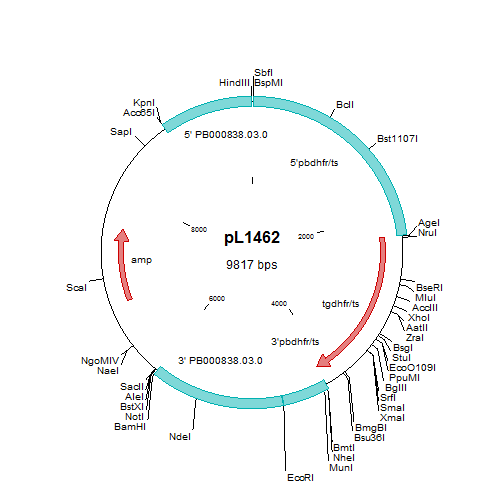
Generation of P. berghei mutants lacking expression of sequestrin (PbΔsequestrin)
A. Schematic representation of the construct pL1462 targeting sequestrin for gene deletion by double cross-over homologous recombination at the target regions (grey boxes), and the locus before and after disruption. The construct contains the tgdhfr-ts selection marker (SM). Two independent mutants were generated: Δsequestrin-a (1424cl1) and Δsequestrin-b (1353cl2). Primer positions for diagnostic PCRs and amplicon sizes are shown (see Table S1 for primer sequences). B. Diagnostic PCRs (left) and Southern analyses of separated chromosomes (right) confirm the correct integration of the constructs in Δsequestrin-a and Δsequestrin-b. Primer pairs and amplicon sizes are shown in A. Selectable marker (M; primers 4501/4502); 5’-integration event (5’; primers 4459/4703); 3’-integration event (3’; primers 4704/4460); ORF (O; primers 4472/4473). See Table S1 for the sequence of the primers. For Southern analyses, pulsed field gel-separated chromosomes were hybridized with a 3’UTR pbdhfr probe. The Δsequestrin-a mutant has been generated in the reference P. berghei ANKA PbGFP-Luccon which has a gfp-luciferase gene integrated into the silent 230p locus (PBANKA_030600) on chromosome 3. Hybridization with the 3’UTR dhfr/ts probe recognizes the integrated construct on chromosome 10, the reporter GFP-Luccon construct on chromosome 3, and the endogenous dhfr/ts gene located on chromosome 7. Δsequestrin-b has been generated in the reference P. berghei ANKA line cl15cy1. Hybridization with the 3’UTR dhfr/ts probe recognizes the integrated construct on chromosome 8 and the endogenous dhfr/ts gene located on chromosome 7. C. RT-PCR analysis showing absence of sequestrin transcripts in liver stages of Δsequestrin-a. PCR amplification using RNA of cultured liver stages (40hpi) was performed either in the presence or absence of reverse transcriptase (RT+ or RT-, respectively) using the primers as shown in the left panel (see Table S1 for the sequence of the primers). The P. berghei hsp70 gene was used as a positive control.
Other mutants
An identical mutant RMgm-(931) was generated in the WT background using the same targeting plasmid as described here. RMgm-799 describes an independent mutant lacking expression of Sequestrin/LISP2
RMgm-593 - A mutant expressing GFP under the control of the promoter of PBANKA_100300 (named lisp2 or sequestrin)
RMgm-800 - A mutant expressing a C-terminal tagged version of PBANKA_100300 (named lisp2 or sequestrin)
|
 *RMgm-930
*RMgm-930 Disrupted: Mutant parasite with a disrupted gene
Disrupted: Mutant parasite with a disrupted gene




 Transgene: Mutant parasite expressing a transgene
Transgene: Mutant parasite expressing a transgene